Apr. 17, 2026
For procurement teams in veterinary pharmaceutical and pet supplement industries, selecting the right prefilled syringe packaging is critical. OverlookedPain points in veterinary prefilled syringe packaging cause avoidable losses. This guide helps identify key pain points and implement practical solutions to reduce risks.
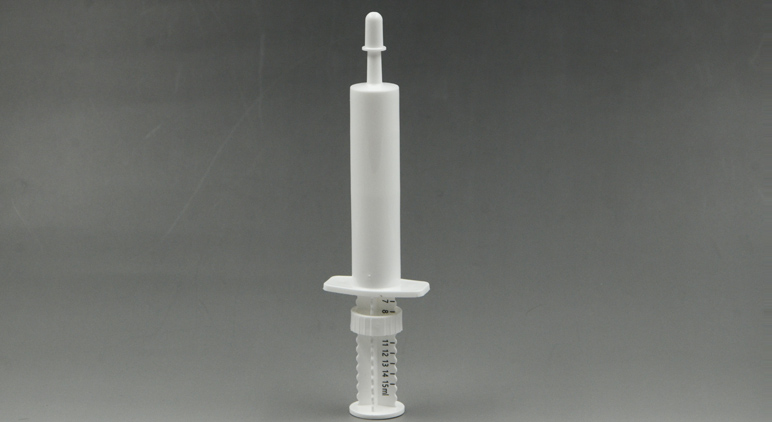
Pain points in veterinary prefilled syringe packaging directly trigger procurement risks: cost risk (batch waste from leaky syringes), compliance risk (failing FDA compliant veterinary prefilled syringe packaging standards), and supply chain risk (production line jams). Teams must focus on these pain points to avoid losses, aligning with how to choose veterinary prefilled syringe supplier to avoid risks.
Pain Point: Leakage in storage/transport; Risks: Batch rejection, contamination, compliance fines;
Check: Vacuum tests, high-quality gaskets (critical for veterinary prefilled syringe for high viscosity paste procurement).
Pain Point: Paste stuck on inner walls; Risks: Raw material waste;
Check: Ultra-smooth inner walls, residue test reports (key for how to choose veterinary prefilled syringe supplier to avoid risks).
Pain Point: Unclear graduations, sticky plungers;
Risks: Ineffective treatment, liability claims; Check: Test dose accuracy (meets FDA compliant veterinary prefilled syringe packaging).
Choose pharmaceutical-grade materials, customize to fit high-viscosity formulations, aligning with how to choose veterinary prefilled syringe supplier to avoid risks.
Previous: None
 冀ICP备11016487号-1
冀ICP备11016487号-1